Clinrap 600 Injection
Price 254 INR/ Vial
Clinrap 600 Injection Specification
- Dosage Form
- Injection
- Origin of Medicine
- India
- Brand Name
- Clinrap 600
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Life Span
- 24 months from manufacturing
- Indication
- Serious Infections due to Susceptible Strains of Microorganisms
- Packaging Type
- Boxed vial
- Salt Composition
- Clindamycin Phosphate
- Drug Type
- Allopathic
- Ingredients
- Clindamycin Phosphate
- Physical Form
- Liquid (Injection)
- Function
- Antibiotic therapy
- Recommended For
- Bacterial Infections
- Dosage
- As directed by physician
- Dosage Guidelines
- Intramuscular/Intravenous use as prescribed
- Suitable For
- Adults
- Quantity
- 2 ml per vial
- Storage Instructions
- Store below 25C, Protect from light
- Regulatory Approval
- WHO-GMP Certified Manufacturing unit
- Therapeutic Class
- Lincosamide Antibiotic
- Contraindications
- Hypersensitivity to clindamycin or lincomycin
- Side Effects
- Diarrhea, abdominal pain, nausea, rash
- Strength
- 600 mg/2 ml
- Appearance
- Clear, colorless to pale yellow solution
- Marketed By
- Rapross Pharmaceuticals
- Route of Administration
- IM/IV
- Manufactured By
- Rapross Pharmaceuticals Pvt. Ltd.
- Prescription Required
- Yes
- Shelf Life
- 24 months
Clinrap 600 Injection Trade Information
- Minimum Order Quantity
- 10 Vials
- Supply Ability
- 1000 Vials Per Month
- Delivery Time
- 2-10 Days
About Clinrap 600 Injection
Clinical Usage and Applications of Clinrap 600 Injection
Clinrap 600 Injection is recommended for the management of serious bacterial infections due to susceptible strains of microorganisms. Its application is intramuscular or intravenous, making it suitable for hospital settings and outpatient care under strict medical supervision. Clinrap 600 is specifically used in adults and is guided by a physician's dosage instructions. The injection delivers fast, effective antibiotic therapy and is ideal for both general and severe infection cases requiring robust intervention.
Sample, Payment, and Supply Details for Clinrap 600 Injection
Samples of Clinrap 600 Injection can be arranged for evaluation. Payment terms are flexible, supporting both domestic market transactions and international orders. Rapross Pharmaceuticals ensures reliable supply ability, with express shipping available for urgent needs. Products are promptly dispatched, typically in boxed vials, to meet diverse distributor, exporter, and supplier requirements. Clinrap 600 is conveniently shipped to multiple locations, ensuring timely delivery to healthcare facilities across India and worldwide.
FAQ's of Clinrap 600 Injection:
Q: How should Clinrap 600 Injection be administered?
A: Clinrap 600 Injection should be administered either intramuscularly or intravenously, strictly following the dosage instructions provided by a healthcare professional.Q: What conditions is Clinrap 600 Injection recommended for?
A: This injection is specifically recommended for serious bacterial infections caused by susceptible strains of microorganisms, as determined by your physician.Q: When should I avoid using Clinrap 600 Injection?
A: Clinrap 600 Injection should not be used if you have a known hypersensitivity to clindamycin or lincomycin. Consult your doctor for alternative treatments if you have these allergies.Q: Where is Clinrap 600 Injection manufactured and by whom?
A: Clinrap 600 Injection is manufactured and marketed by Rapross Pharmaceuticals Pvt. Ltd., in a WHO-GMP certified facility in India.Q: What is the process for storing Clinrap 600 Injection?
A: Store this product below 25C and protect it from light. Follow all instructions on the packaging for optimal safety and effectiveness.Q: What are the possible side effects of Clinrap 600 Injection?
A: Common side effects may include diarrhea, abdominal pain, nausea, and rash. Notify your doctor if you experience persistent or severe symptoms.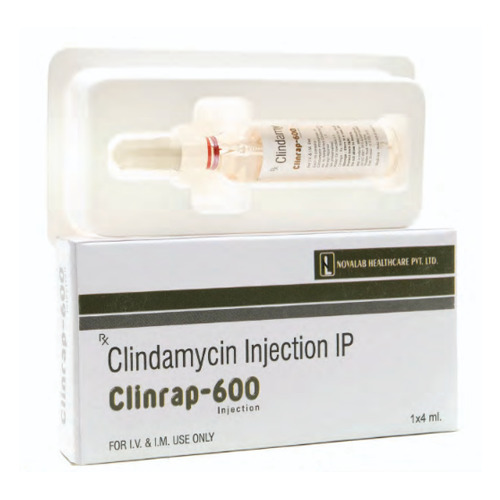

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Injection Category
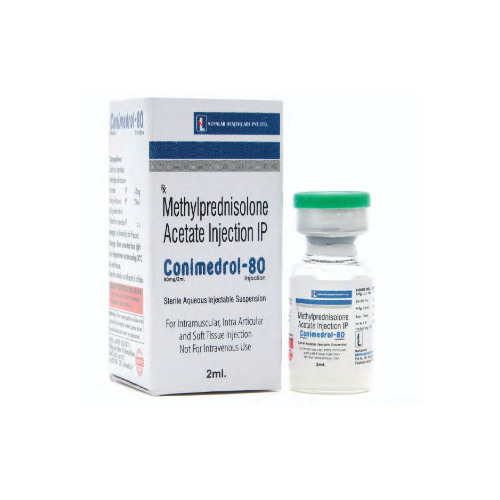
Conimedrol 80 Injection
Price 102.27 INR / Vial
Minimum Order Quantity : 10 Vials
Storage Instructions : Store below 25C, protect from light and moisture
Recommended For : Antiinflammatory and immunosuppressive therapy
Ingredients : Methylprednisolone Sodium Succinate 80 mg
Physical Form : Other, Injection
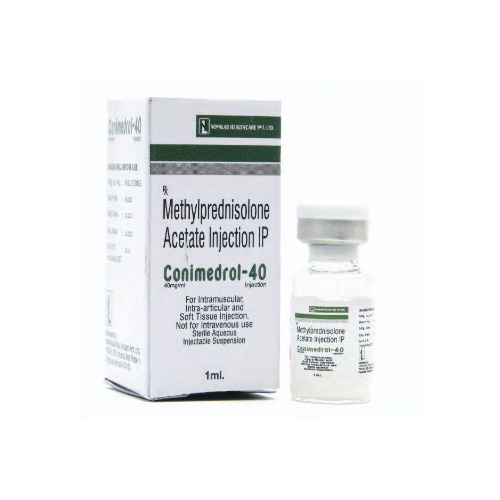
Conimedrol 40 Injection
Price 51.13 INR / Vial
Minimum Order Quantity : 10 Vials
Storage Instructions : Store below 25C, protect from light and moisture
Recommended For : Antiinflammatory and immunosuppressive therapy
Ingredients : Methylprednisolone Acetate
Physical Form : Other, Injection
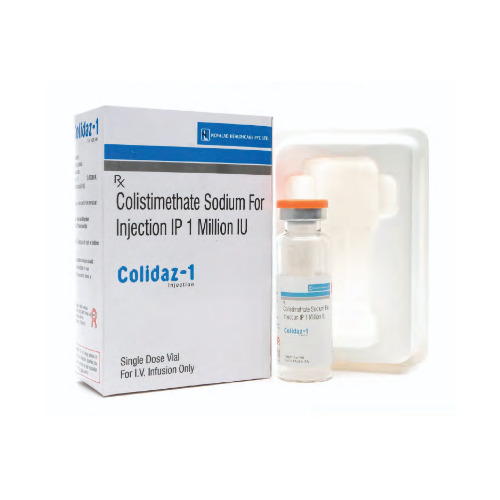
Colidaz 1 Injection
Price 925 INR / Vial
Minimum Order Quantity : 1 Vial
Storage Instructions : Store in a cool and dry place, Protect from light
Recommended For : Bacterial Infections, HospitalAcquired Infections, Treatment under medical supervision
Ingredients : Cefoperazone 1000mg + Sulbactam 500mg
Physical Form : Other, Liquid/Injection
Colidaz 2 Injection
Price 1475 INR / Vial
Minimum Order Quantity : 1 Vial
Storage Instructions : Store in a cool, dry place below 25C. Protect from light.
Recommended For : Bacterial Infections
Ingredients : Cefoperazone 1000mg + Sulbactam 1000mg
Physical Form : Other, Injection


 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese